April 2026 TPJ Editor choice: Hidden highways: Mapping plasmodesmata in Arabidopsis roots
Highlighting: https://doi.org/10.1111/tpj.70726
Hidden highways: Mapping plasmodesmata in Arabidopsis roots
Cytoplasmic connections between plant cells were discovered more than hundred years ago by the botany professor Eduard Tangl. He hypothesised that these connections serve as communication and transport pathways between cells. Today, these connections are called plasmodesmata (PDs). PDs are typically found in clusters known as pit fields and located in thinner regions of the cell wall. Their abundance and distribution influence the transport of molecules between cells. Therefore, understanding the 3D cell type-specific spatial patterning of PDs supports our understanding of how potential routes of direct cell communication are established in plant organs.
Confocal microscopy provides a rapid method for mapping pit field distribution; however, this approach has so far been limited to the most accessible outer portions of living leaf tissue. To overcome this limitation, Davis et al. used confocal microscopy in combination with cleared tissue and fluorescent reporters to map pit field distribution in Arabidopsis roots. From that, they developed a quantitative approach for mapping plasmodesmata pit fields across roots in 3D, at cell type- and cell interface-specific resolution.
First, the authors imaged PD pit fields and used fluorescence intensity above a defined threshold as a proxy to estimate the abundance of PD pit fields at cell interfaces. The analyses were performed across all root cell types and along both radial and apical–basal axes.
The authors detected the highest total signal at apical–basal cell interfaces, consistent with earlier observations. They hypothesised that this spatial arrangement of PDs facilitates rapid symplasmic transport along root cell files and along the developmental axis of the root. The enrichment of PDs at apical–basal interfaces may also reflect the need for directional transport of proteins and hormones that maintain developmental gradients. Notably, apical–basal interfaces correspond to the planes of cell division and elongation in the root. Primary PDs are formed during cytokinesis, which may partly explain their higher abundance at these interfaces. In addition, the insertion of secondary plasmodesmata during cell elongation may further contribute to this pattern.
During development, the total signal increased at the endodermal cell interfaces, while proportionally decreasing at outer cell interfaces. Similarly, the signal density of pit fields increased at endodermal interfaces as the root developed. These patterns suggest that PD distribution is not static but changes with developmental stage, as reflected by differences between the meristem and transition zone. This shift may reflect pre-patterning associated with Casparian strip formation. In Arabidopsis roots, the Casparian strip forms within the endodermis, and the strip already functions as an effective symplasmic barrier near the meristem, within the transition zone and early elongation zone.
In terms of PD distribution, the authors observed more clustering of PDs into pit fields at cortex–endodermis radial interfaces in the transition zone compared with the meristem. In contrast, on endodermal apical–basal interfaces, PD were less clustered into pit fields in the transition zone than in the meristem. The authors hypothesise that this pattern may reflect increased de novo formation of secondary PDs at these interfaces during development.
Brassinosteroids have been shown to influence the opening and closing of PDs; however, it remains unclear whether these hormones also affect PD abundance and spatial distribution. To investigate this, the authors analysed PD occurrence and distribution in the brassinosteroid insensitive 1 (bri1) mutant, which exhibits a diminished response to brassinosteroids. Quantitative analysis showed relative increases in both total signal and signal density on radial cell walls, alongside a relative decrease in signal density at the apical–basal interface of the epidermis. Spatial pattern analysis further showed an increase in Moran’s I values in bri1, indicating greater uniformity in PD patterning. These results suggest that BRI1 plays a role in regulating PD distribution within pits.
In the future, the authors intend to map PD distribution throughout plant tissues to investigate cell communication at organ level, and how this can change in response to the environment. This approach paves the way to understand the intracellular connections of plants and the complex decision-making processes essential to plant survival in an ever-changing world.
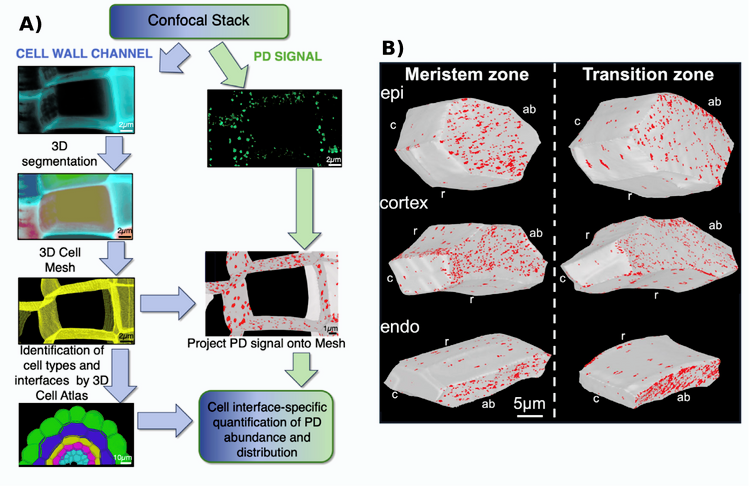
Figure 1: Cell type-specific plasmodesmata (PD) distribution in Arabidopsis.
A) Workflow for quantification of PD pit field distribution in 3D using confocal microscopy.
B) Pit field distribution in the meristematic zone and in the transition zone. UBQ10::SP-mCherry-PDCB1 signal indicating PD pits is projected onto cell surfaces for epidermis (epi), cortex and endodermis (endo) cells; cell interface orientation apical-basal (ab), circumferential (c), and radial (r). Modified from (Davis et al., 2026).






