JXB Volume 74, Issue 14 – Editor’s choice
This article highlights the following publication:
Jourquin J, Fernandez AI, Wang Q, Xu K, Chen J, Šimura J, Ljung K, Vanneste S, Beeckman T. 2023. GOLVEN peptides regulate lateral root spacing as part of a negative feedback loop on the establishment of auxin maxima. Journal of Experimental Botany 74, 4031–4049.
https://doi.org/10.1093/jxb/erad123
You say “Yes”, I say “No”, you say “Stop” and I say “Go, go, go!”
The Beatles knew very well that opposites don’t just make catchy pop songs, as proven in their hit single “Hello Goodbye”, they actually depend on and complement each other. It was the sweet and salty combination of Paul McCartney’s cheerful optimism and John Lennon’s contemplative surrealism that propelled the band to global fame and made the two men the most successful songwriting duo in music history, with McCartney describing their relationship as a “very friendly competition”.
The concept of duality in which opposites work closely together also applies to the regulation of plant root system architecture, as revealed by Jourquin et al. (2023) in issue 14 of the Journal of Experimental Botany. The team found that activators and inhibitors of lateral root initiation act hand in hand in the same group of cells to determine the outgrowth of lateral roots along the primary root axis.
The activator in this case is the phytohormone auxin, undoubtedly the most studied of all plant hormones. Auxins are, in fact, a group of chemical compounds with indole-3-acetic acid (IAA) being the most abundant endogenous form in plants. Originally named for their effect to promote plant growth (the Greek word “auxein” means “to grow”), over 100 years of intense research has shown that auxins are ubiquitously involved in numerous developmental processes, described in detail by Caumon and Vernoux (2023) in their recent JXB Darwin Review. Charles Darwin was, incidentally, one of the first to describe auxin in action in his 1880 published book “The Power of Movement in Plants”. He marvelled at the capacity of roots to react to environmental stimuli such as light, moisture and gravity – responses that are all mediated by auxin. From his carefully conducted and meticulously recorded experiments he concluded that external signals are sensed and integrated by a group of cells in the root tip, which then “transmits some influence or stimulus to the adjoining parts, causing them to bend to or from the source of irritation”. We now know that this transmitter is auxin and that not only root movement but also the positioning of lateral roots is determined by auxin signals emanating from the root tip. However, our understanding of the exact molecular mechanisms underlying lateral root spacing and development has thus far been incomplete.
Development of lateral roots starts in the root pericycle, a layer of cells surrounding the vasculature. Auxin signals that originate in regular time intervals of a few hours in the root tip have been found to render certain pericycle cells along the main root axis competent for future lateral root formation. Increased auxin levels in these periodically primed lateral root founder cells (LRFCs) cause an asymmetric perpendicular cell division that marks the onset of further root development. However, not all primed LRFCs give rise to new lateral roots and previous studies have shown that small signalling peptides of the GOLVEN (GLV) family may be involved in inhibiting lateral root initiation. The importance of peptides as chemical messengers between plant cells has long been underestimated because of intense focus on few phytohormones and since their small size of usually <20 amino acids and oftentimes low expression levels rendered them “invisible” in genomic studies. However, increased research over the past two decades or so shows that signalling peptides are essential for cell-to-cell communication and involved in various processes throughout a plant’s life. The research group of Tom Beeckman focusses on their role in root development and found that plants overexpressing GLV6 or GLV10 have significantly fewer lateral roots (Fernandez et al., 2013). Interestingly, expression of GLV6/10 is auxin inducible and their expression is up-regulated in LRFCs (Fernandez et al., 2015, 2020). The team set out to bring some clarity to this counterintuitive simultaneous accumulation of lateral root activators and repressors in the same cells, yet their gene expression analysis yielded more surprising results: the vast majority of genes upregulated by GLV6/10 is also up-regulated by auxin, while the vast majority of genes down-regulated by GLV6/10 is also down-regulated by auxin (Jourquin et al., 2023). The most straightforward explanation for this large overlap in transcriptional responses would be that GLV6/10 activity is established via auxin. However, the team could demonstrate in different mutants that GLV6/10 exert their effect independent of auxin perception and signalling. Importantly, GLV6/10 only regulate a subset of all auxin controlled genes and it is presumed that the partial activation of the auxin response by GLV6/10 leads to pericycle cell divisions that are non-formative and do not give rise to lateral roots. Furthermore, high levels of GLV6/10 hamper auxin accumulation in LRFCs presumably through increased expression of auxin transporters that promote auxin efflux. Based on their results the authors propose a model that describes how the balanced interplay between activating auxin and inhibiting GLV peptides determines lateral root initiation and spacing (Figure 1): Auxin accumulation in LRFCs induces expression of GLV6/10 peptides that are secreted into the apoplast. This secretion, in combination with positive feedback mechanisms that further increase local auxin levels, results in a high cellular auxin/GLV ratio and allows for lateral root initiation and growth. GLV6/10 peptides diffuse through the apoplast creating a concentration gradient around the initial pair of LRFCs where auxin accumulation in pericycle cells is impaired, most probably via the GLV-induced increase in auxin efflux transporters. Lateral root initiation is consequently stalled and development of neighbouring lateral roots can only be initiated in zones where GLV6/10 levels are low enough to allow for sufficient auxin accumulation in LRFCs.
The combination of systematic priming of potential pre-branch sites and additional fine-tuning of lateral root initiation via cell-to-cell signalling allows plants to adjust root system architecture to nutrient and water demand and to quickly respond to changing environmental conditions. The new understanding of the “very friendly competition” between lateral root activators and inhibitors presented in Jourquin et al. may help to develop plants with higher water and nutrient uptake efficiency and better stress resistance in challenging environments.
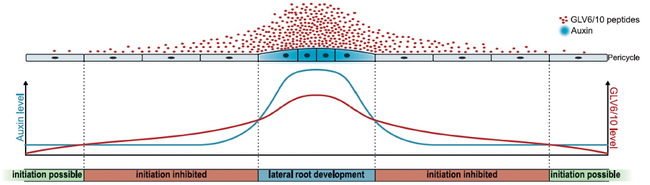
Figure 1: Proposed model for the regulation of lateral root initiation and spacing via the balance between auxin and GLV6/10 peptide levels (Jourquin et al., 2023).
References
Caumon H, Vernoux T. 2023. A matter of time: auxin signaling dynamics and the regulation of auxin responses during plant development. Journal of Experimental Botany 74, Issue 14, 3887–3902.
Darwin, C. R. 1880. The power of movement in plants. London: John Murray.
Fernandez A, Drozdzecki A, Hoogewijs K, Nguyen A, Beeckman T, Madder A, Hilson P. 2013. Transcriptional and functional classification of the GOLVEN/ROOT GROWTH FACTOR/CLE-like signaling peptides reveals their role in lateral root and hair formation. Plant Physiology 161, 954–970.
Fernandez A, Drozdzecki A, Hoogewijs K, Vassileva V, Madder A, Beeckman T, Hilson P. 2015. The GLV6/RGF8/CLEL2 peptide regulates early pericycle divisions during lateral root initiation. Journal of Experimental Botany 66, 5245–5256.
Fernandez AI, Vangheluwe N, Xu K, et al. 2020. GOLVEN peptide signalling through RGI receptors and MPK6 restricts asymmetric cell division during lateral root initiation. Nature Plants 6, 533–543.





